 ISO 14155-1:2003, Clinical investigation of medical devices for human subjects - Part 1: General requirements.ISO 13683:1997, Sterilization of health care products - Requirement for validation and routine control of moist heat sterilization in health care facilities.ISO 13641:2002 Part 1 – General test & ISO 13641:2002 Part 2 – Test for low biomass concentrations, Elimination or reduction of risk of infection related to in vitro diagnostic medical devices.ISO 11137:1995, Sterilization of health care products - Requirements for validation and routine control - Radiation sterilization (Corrigendum 1 published 1995 Amendment 1 published 2001).ISO 11135:1994, Medical devices - Validation and routine control of ethylene oxide sterilization (Corrigendum 1 published 1994).ISO 11134:1994, Sterilization of health care products - Requirements for validation and routine control - Industrial moist heat sterilization.ISO 10012, Measurement management systems - Requirements for measurement processes and measuring equipment.ISO 9001:2000, Quality management systems - Requirements.Here is a list of the standards referenced in ISO 13485 bibliography § ISO 14971:2007 Medical devices Application of risk management to medical devices § ISO Guide 73 - 2009 - Risk management - Vocabulary.  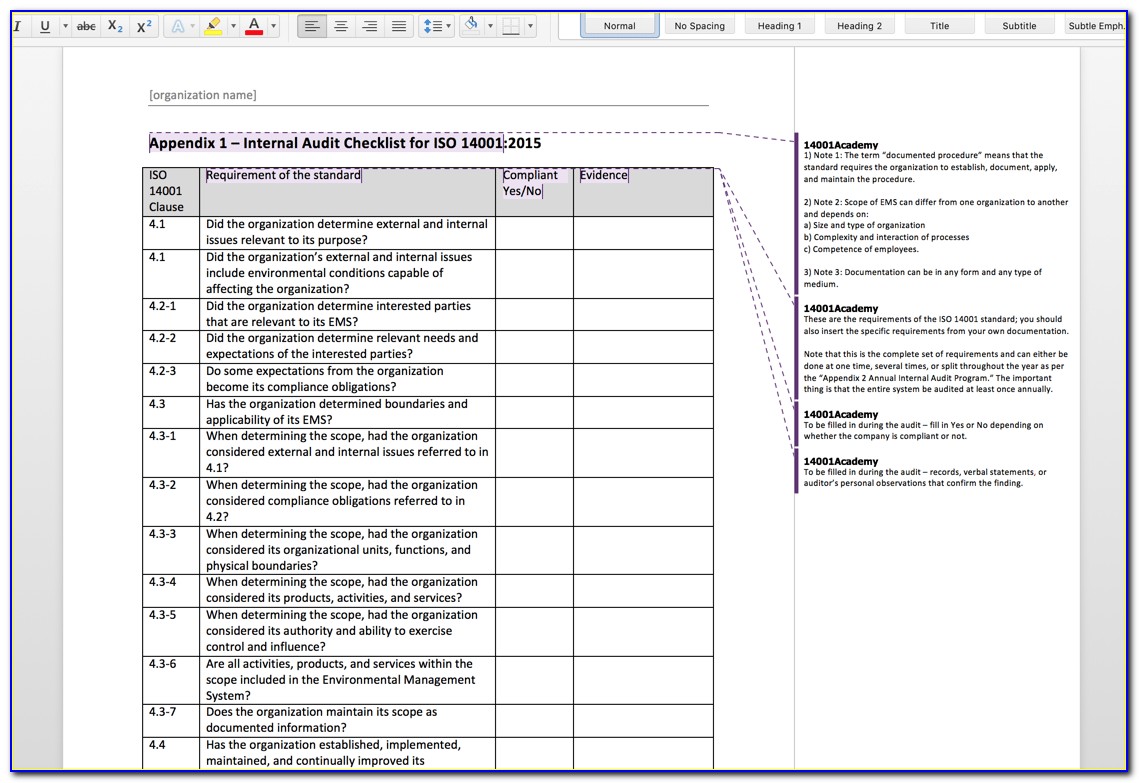
Risk Management is a requirement-Product Realization clause 7.1 o See guidance standards.Buy copies of the ISO13485 standard to pinpoint the areas that need attention. 
Comparison between ISO 9001-and-ISO-13485.Links to buy Standards directly from the source are Underlined Bold Red text Here are some resources:.Links to supporting information are underlined blue text.Requirements in addition to ISO 9001 are Highlighted in yellow in this document Throughout this document, you will find the following assistance:. 
ISO 13485:2003 - INTERNAL AUDIT CHECKLIST = + – 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
